Photochromic dithienylethenes characterized by in situ irradiation NMR-spectroscopy and electrochemically induced responsiveness on gold substrates†
Abstract
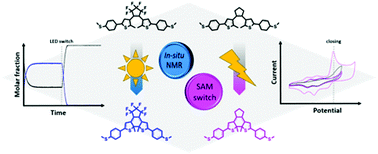
Photochromic compounds comprising small molecules and polymers have been shown to be of great interest in the field of stimuli-responsive materials for applications as sensors and (nano-) devices. In this regard dithienylethenes (DTEs) have been proven to be especially valuable because of their tolerance to chemicals and their good stability making them tunable and reversibly switchable between an open and a closed structure featuring different optical properties. By utilizing a potent in situ irradiation NMR spectroscopy approach, it is possible to determine the photochromic switching capability of two DTE derivatives in solution leading to crucial information about the structures, the equilibria of the chemical ring-opening and closing of the DTEs as well as the longtime stability of these molecular switches. In particular the perfluorinated thioether bearing compound shows remarkable stability with little fatigue related to the irreversibly cyclized byproduct known for DTEs. The thioether-bearing DTEs can be deprotected and used for immobilization on gold substrates which are characterized by water contact angle measurements prior to and after DTE functionalization. Finally, the electrochemically induced switching capability of the perhydro DTE is investigated by cyclic voltammetry proving its fast, quantitative and reversible cyclization. We envisage these materials for applications as sensors and optical switching devices.


 Please wait while we load your content...
Please wait while we load your content...