Phase transition, optical and dielectric properties regulated by anion-substitution in a homologous series of 2D hybrid organic–inorganic perovskites†
Abstract
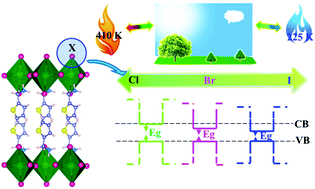
Functional regulation is a key issue for the application of two-dimensional (2D) hybrid organic–inorganic perovskites in the fields of ferroelectrics, photovoltaics and fluorescence, etc. Here, we report a homologous series of new 2D hybrid organic–inorganic halide perovskites [(C4H3S)CH2NH3]2PbX4 ((C4H3S)CH2NH3+ = thienylmethylamine; X = Cl−, Br−, I−). Their properties, including crystal structures of high (HT) and low (LT) temperature phases, phase transition temperature (Tc), and dielectric constants (ε′), exhibit significant discrepancies as the halogen atoms vary from Cl, to Br to I. This effect was unveiled to be divergent hydrogen bond interactions between the organic (C4H3S)CH2NH3+ cation and the inorganic [PbX6]2− octahedron layer resulting from the different radii of the halogen ions. Meanwhile, the direct band gaps of [(C4H3S)CH2NH3]2PbX4 were tuned from 3.78 eV (X = Cl−), to 3.08 eV (X = Br−) to 1.99 eV (X = I−) with a large regulative magnitude of 1.79 eV. The photoluminescence (PL) emission wavelength increases from 505, to 526 to 545 nm for Cl, Br and I, respectively, with the full width at half-maximum (FWHM) significantly decreasing from 123, 69 to 41 nm due to the quantum size effect and structural distortion. However, the average lifetimes of the three compositions reveal the reverse trend of Br > Cl > I, which is attributed to the changes of hydrogen bonds between organic [(C4H3S)CH2NH3]+ cations and inorganic PbX6 octahedral layers and thermal vibration. These results display the high tunability and versatility of the 2D hybrid organic–inorganic perovskites.


 Please wait while we load your content...
Please wait while we load your content...