Insight into in-plane isotropic transport in anthracene-based organic semiconductors†
Abstract
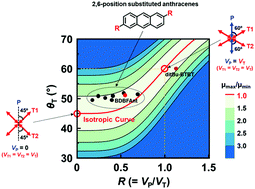
We report a new insight into the molecular design strategy for developing anthracene derivatives with in-plane isotropic mobility. Among three types of anthracene derivatives: 2-,9,10-, and 2,6-positions substituted anthracenes (2-ANTs, 9,10-ANTs, and 2,6-ANTs), we discovered that 2,6-ANTs offer the highest degree of in-plane isotropy in mobility. By analyzing the structure and properties of 2,6-ANTs, we identified conditions to achieve in-plane isotropic mobility: first, the molecular arrangement of the layered herringbone (LHB) structure needs to be symmetrical with respect to both the π–π stacking direction and the transverse direction. Second, the angle between the π–π stacking direction and the transverse direction, θT, needs to be around 50°. Third, the ratio between the transfer integrals along the π–π stacking and transverse directions, R = VP/VT, needs to be smaller than 0.7. Therefore, both structural features and electronic property parameters determine the degree of isotropy. An investigation on the torsion angles between the side chain and the anthracene backbone found that a desirable R value could be obtained by improving the planarity in 2,6-ANTs to enhance the intermolecular interactions on the transverse contacts. To confirm the theory, 2,6-bis(dibenzo[b,d]furan-3-yl)anthracene (BDBFAnt) with fused aromatic rings as substituents was used to form a single-crystal organic field effect transistor (SC-OFET) and it demonstrated an isotropic mobility with values within a range of 2.6–3.4 cm2 V−1 s−1 in all measured directions.


 Please wait while we load your content...
Please wait while we load your content...