A novel material for the detection and removal of mercury(ii) based on a 2,6-bis(2-thienyl)pyridine receptor†
Abstract
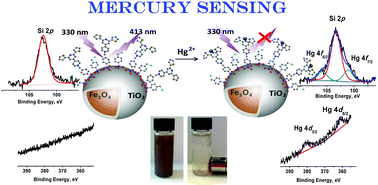
Here we report on the strong potential for mercury Hg(II) detection and uptake demonstrated by a 2,6-bis(2-thienyl)pyridine molecular receptor (L). The coordination affinity of L with Hg(II) was studied experimentally and computationally. Based on the results of 1H NMR and Density Functional Theory (DFT), L binds to Hg(II) via SNS chelation. L demonstrates a strong ability to perform as a single excitation (330 nm)–single emission (413 nm) selective “turn off” fluorimetric sensor for Hg2+ ions. Furthermore, time-dependent DFT calculations predicted and experimental studies confirmed the ability of this system to act as a single excitation (385 nm)–dual emission ratiometric Hg(II) sensor with two well-defined emission bands, “turn off” at 413 nm and “turn on” at 563 nm. Through detailed studies at both emission wavelengths (413 and 563 nm) we show the ability of the ligand to effectively detect mercury ions and demonstrate the potential of the system to differentiate Hg2+ from Zn2+, Cd2+, Cu2+, Cr3+, Co2+, Ru3+, and Fe2+ ions with minimum to no interference. Chemical anchoring of L via siloxane chemistry on surface-enhanced magnetite/titania core–shell nanospheres results in a novel material with strong potential for Hg(II) uptake from aqueous and organic solutions. The magnetite core allows fast removal of the nanoparticles after the reaction by applying an external magnetic field. The resulting nanomaterial demonstrates promising performance as a single excitation (330 nm)–single emission (413 nm) sensor for Hg2+ ions. The presence of iron ions in analytical matrices leads to interference, influencing the fluorimetric detection of Hg2+. To address this challenge, the material can be deposited on a glassy carbon electrode where L-complexed iron and mercury ions can be electrochemically resolved.


 Please wait while we load your content...
Please wait while we load your content...