The effect of a heavy atom on the radiative pathways of an emitter with dual conformation, thermally-activated delayed fluorescence and room temperature phosphorescence†
Abstract
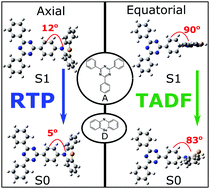
This study focuses on the photophysics of a donor–acceptor molecule with 2,4,6-triphenyl-1,3,5-triazine and phenoselenazine acceptor and donor units. Selenium distinctively folds the phenoselenazine resulting in the appearance of two conformers, yielding both room temperature phosphorescence (RTP) and thermally-activated delayed fluorescence (TADF). This combination produces a unique in-depth picture of the mechanisms inherent to dual emission in TADF molecules. We revisit the photophysics of the phenothiazine analogue and observe similar behaviours. The dual emission comes from a short-lifetime quasi-axial conformer and longer-lifetime quasi-equatorial conformer giving efficient TADF. The quasi-axial conformer is not TADF active because it has a low energy local triplet that quenches the charge transfer states, giving rise to RTP. From sulphur to selenium, we point to an enhancement in the phosphorescence radiative decay with little effect on the reverse intersystem crossing (rISC) which means that adding a heavy atom to the donor does not affect the TADF performance. The interplay between TADF and RTP leads to yellow-green emitting devices with external quantum efficiencies of 17% from the optimisation of the conformer that gives rise to TADF. Therefore, the subtle effects enhanced by the phenoselenazine donor further add to our knowledge about how the molecular structure affects TADF photophysics.


 Please wait while we load your content...
Please wait while we load your content...