Rational design of thermally activated delayed fluorescence emitters with aggregation-induced emission employing combined charge transfer pathways for fabricating efficient non-doped OLEDs†
Abstract
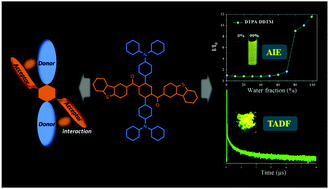
The development of luminescent materials simultaneously possessing thermally activated delayed fluorescence (TADF), aggregation-induced emission (AIE), and high luminescence efficiency in the solid state is urgent for fabricating efficient non-doped organic light-emitting diodes (OLEDs). Herein, two novel emitters bearing triphenylamine as the donor moiety and phenyl ketone as the acceptor unit, named as DTPA-DTM and DTPA-DDTM, were designed and synthesized. Combined charge transfer pathways in a single molecule could be achieved via linking the donor and acceptor units in the ortho-position, which was favorable for reducing singlet–triplet energy splitting (ΔEst) and increased the luminescence efficiency. The TADF characteristics, AIE properties, solvatochromism, theoretical calculations, and crystallography of the designed emitters were systematically investigated to determine the structure–property relationship. By virtue of the inhibited concentration quenching in the solid state, the non-doped OLEDs employing DTPA-DDTM as the emitter realized high current efficiency (LE) and external quantum efficiency (EQE) of 25.6 cd A−1 and 8.2%, respectively. The obtained results clearly demonstrate that combining the charge transfer pathways in a single molecule is a promising strategy to construct highly efficient TADF emitters that can be used for fabricating non-doped organic light-emitting diodes.


 Please wait while we load your content...
Please wait while we load your content...