Modulation of the conductance in platinum(ii) bis(acetylide) molecules through “gating” metal ions†
Abstract
Mononuclear platinum(II) complex trans-Pt(PPh3)2(C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CC6H4SMe-4)2 (1-Pt) and its derivatives [PtM(dppm)2(C
CC6H4SMe-4)2 (1-Pt) and its derivatives [PtM(dppm)2(C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CC6H4SMe-4)2]+ (dppm = bis(diphenylphosphino)methane; M = Au, 2-PtAu; Ag, 2-PtAg; Cu, 2-PtCu) with “mono-gating” metal ions and [PtM2(dpmp)2(C
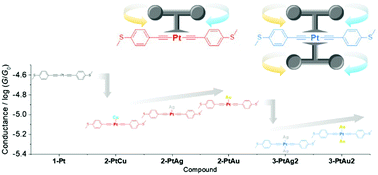
CC6H4SMe-4)2]+ (dppm = bis(diphenylphosphino)methane; M = Au, 2-PtAu; Ag, 2-PtAg; Cu, 2-PtCu) with “mono-gating” metal ions and [PtM2(dpmp)2(C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CC6H4SMe-4)2]2+ (dppm = bis(diphenylphosphinomethyl)phenylphosphine; M = Au, 3-PtAu2; Ag, 3-PtAg2) with “dual-gating” metal ions were elaborately designed to modulate the conductance in platinum(II) bis(σ-acetylide) complexes through “gating” d10 metal ions. The single-molecule conductance measurements by the STM-BJ technique demonstrate that the conductance follows 10−4.5 (1-Pt) > 10−4.8 (2-PtAu) > 10−4.9 (2-PtAg) > 10−5.0 (2-PtCu) > 10−5.1 (3-PtAu2) > 10−5.3 (3-PtAg2) G0. Undoubtedly, charge transport along the platinum(II) bis(σ-acetylide) backbone is progressively restricted by introducing one to two “gating” M ions to the mononuclear platinum(II) molecule. As revealed by X-ray crystallography, the “gating” d10 metal ion is substantially bound to the acetylide and in fact the M–acetylide bonding is progressively enhanced in the order Au–Cacetylide → Ag–Cacetylide → Cu–Cacetylide, which coincides with a progressive decrease in the conductance as 10−4.8 (2-PtAu) > 10−4.9 (2-PtAg) > 10−5.0 (2-PtCu) G0. The M–acetylide coordination blocks the charge transport along the platinum(II) bis(σ-acetylide) backbone so that the d10 metal ion behaves as a resistor to suppress the conductance. Theoretical studies confirm that the “gating” metal ions control the HOMO energy, which is lowered by the increased d orbital contribution of “gating” metal ions through Pt–M interactions.
CC6H4SMe-4)2]2+ (dppm = bis(diphenylphosphinomethyl)phenylphosphine; M = Au, 3-PtAu2; Ag, 3-PtAg2) with “dual-gating” metal ions were elaborately designed to modulate the conductance in platinum(II) bis(σ-acetylide) complexes through “gating” d10 metal ions. The single-molecule conductance measurements by the STM-BJ technique demonstrate that the conductance follows 10−4.5 (1-Pt) > 10−4.8 (2-PtAu) > 10−4.9 (2-PtAg) > 10−5.0 (2-PtCu) > 10−5.1 (3-PtAu2) > 10−5.3 (3-PtAg2) G0. Undoubtedly, charge transport along the platinum(II) bis(σ-acetylide) backbone is progressively restricted by introducing one to two “gating” M ions to the mononuclear platinum(II) molecule. As revealed by X-ray crystallography, the “gating” d10 metal ion is substantially bound to the acetylide and in fact the M–acetylide bonding is progressively enhanced in the order Au–Cacetylide → Ag–Cacetylide → Cu–Cacetylide, which coincides with a progressive decrease in the conductance as 10−4.8 (2-PtAu) > 10−4.9 (2-PtAg) > 10−5.0 (2-PtCu) G0. The M–acetylide coordination blocks the charge transport along the platinum(II) bis(σ-acetylide) backbone so that the d10 metal ion behaves as a resistor to suppress the conductance. Theoretical studies confirm that the “gating” metal ions control the HOMO energy, which is lowered by the increased d orbital contribution of “gating” metal ions through Pt–M interactions.


 Please wait while we load your content...
Please wait while we load your content...