Modulating charge transport characteristics of bis-azaisoindigo-based D–A conjugated polymers through energy level regulation and side chain optimization†
Abstract
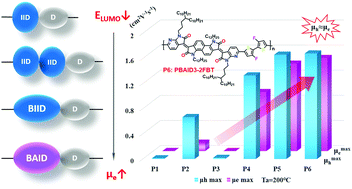
Six donor–acceptor (D–A) conjugated polymers, P1–P6, based on the novel electron acceptors bis-isoindigo (BIID) and bis-azaisoindigo (BAID), were designed and synthesized for solution-processed organic field-effect transistors with high-performance optical and electrical properties. Energy level regulation was achieved by incorporation of nitrogen and fluorine heteroatoms and side chain optimization to reduce side chain density and extend branching point positions. Both the hole (μh) and electron mobilities (μe) significantly increased from P1 to P6, and there was an obvious transition from hole-dominated ambipolar charge transport behavior to highly-balanced ambipolar behavior. Since aza-substitution strengthened the electron-deficient property of the acceptor units, the entire molecular skeleton of BAID-based polymers has a high electron affinity and a low-lying lowest unoccupied molecular orbital (LUMO) level according to ultraviolet photoelectron spectroscopy (UPS) and cyclic voltammetry (CV) measurements. The μe of BAID-based polymers is an order of magnitude higher than that of BIID-based polymers. In addition, side chain optimization was shown to significantly influence intermolecular interactions and supramolecular self-assembly. The stepwise reduction of the π–π stacking distances of P1–P6 calculated by GIXRD curves supports this conclusion. Finally, in P6 (PBAID3-2FBT), a maximum absorption wavelength (λmax) was observed at 797 nm, a low optical band gap (Eg) of 1.27 eV, a low LUMO level of −3.90 eV, and an average μh and μe of 1.31 cm2 V−1 s−1. This paper offers an available molecular design strategy to achieve highly-balanced and high-performance ambipolar charge carrier transport D–A conjugated polymers.


 Please wait while we load your content...
Please wait while we load your content...