Solvent-induced aggregation based on a heteroleptic Ir(iii) complex via hydrogen bonds†
Abstract
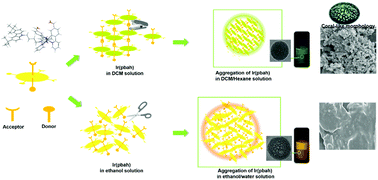
It has become an interesting field to obtain an unambiguous mechanism according to the structure–property relationship. Herein, an unprecedented strategy of molecule design for controlling nano-aggregated size and morphology by assembled hydrogen bonds was shown. A new Ir(III) complex was designed and synthesized. Notably, the functional groups aldehyde, tert-butyl carbazole, and alkyl are critical to its properties. The emission spectra showed dual emissions and each peak had vibrating structures in non-protonic DCM, but not in a protonic ethanol solution, which was assigned to different intermolecular interactions and excitation states in nonpolar and polar solvents. The marked differences of the excitation spectra peaks observed from the high and low energy bands proved the different origins of emission. Furthermore, the concertation-dependent emission for DCM and ethanol solution, and 2D NEOSY NMR, exhibited that the intermolecular hydrogen coupling in space promoted the intermolecular hydrogen bonds (C–H⋯O, C–H⋯π). In addition, while the aggregation state in DCM/hexane solution also showed a fine emission vibrating structure, that in the ethanol/water solution was without that. The DLS and TEM results indicated that the vibrating structures were related to the hydrogen bonds driving aggregated nanoparticle size and shape. SEM also showed their assembled morphology. All results indicated that the hydrogen coupling played a decisive role in aggregating self-assembly or the molecule packing state. These design ideas could provide guidance for novel functionalized nanomaterials.


 Please wait while we load your content...
Please wait while we load your content...