Controllable luminescence of a Li–Al layered double hydroxide used as a sensor for reversible sensing of carbonate†
Abstract
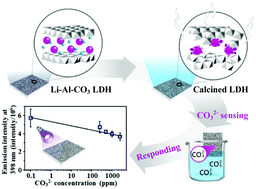
This investigation finds that a Li–Al layered double hydroxide (Li–Al–CO3 LDH) that has no intercalated chromophore and no rare earth dopant could function as a luminescent material with controllable luminescence properties. The luminescence of the LDH can be tuned to ultraviolet (UV), blue or red simply by calcining the Li–Al–CO3 LDH at high temperatures. The LDH calcined at 300 °C emitted strong blue luminescence with its blue emission intensity five times higher than that of the Li–Al–CO3 LDH. The Li–Al–CO3 LDH and the 300 °C-calcined LDH were investigated by 27Al magic angle spinning nuclear magnetic resonance (MAS NMR) spectroscopy. The luminescence mechanism of the strong blue emission is mainly associated with the oxygen defects and pentahedrally-coordinated AlO5 sites in the 300 °C-calcined LDH. Fourier-transform infrared spectroscopy (FT-IR) is a versatile technique to recognize the luminescence intensity of an LDH material. It is because there is a negative relationship between the blue emission intensity from an LDH material and the FTIR intensity of the ν3 mode CO32− adsorption band in the LDH. The functional application of the 300 °C-calcined LDH material was explored and it was found to be an effective luminescent anion sensor that responds to CO32− anions in aqueous solution. The selective sensing of CO32− anions extends over a wide range of concentrations from 0 to 2500 ppm. The 300 °C-calcined LDH exhibits effective reversible sensing of CO32−. The LDH can also be used to detect the furnace temperature during post-processing heat treatment.


 Please wait while we load your content...
Please wait while we load your content...