Kinetic condition driven phase and vacancy enhancing thermoelectric performance of low-cost and eco-friendly Cu2−xS†
Abstract
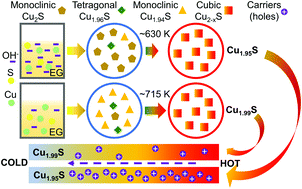
Low-cost and eco-friendly Cu2S has attracted wide research interest in recent years. Here, we synthesized Cu2−xS via a facile solvothermal method. The thermoelectric performance of the as-sintered Cu2−xS can be easily tuned via kinetic-condition control due to subsequently changed phase contents. Based on detailed characterization, it is found that reducing the NaOH amount can preferentially boost the formation of tetragonal Cu1.96S and monoclinic Cu1.94S phases instead of monoclinic Cu2S phase. This phase content change can promote the formation of Cu vacancies in the high-temperature cubic Cu2−xS after the phase transition at ∼700 K. The enhanced Cu vacancy levels can effectively enhance both the power factor (S2σ) and figure of merit (zT) due to simultaneously optimized hole concentration (nH) and reduced phase transition temperature. A peak zT of ∼1.1 is achieved in the Cu2−xS synthesized with 2 ml NaOH with an average composition of Cu1.95S. An average zT of as high as ∼0.76 (T = 573–833 K) is obtained due to both optimized nH leading to enhanced S2σ and reduced phase transition temperature (at ∼700 K).


 Please wait while we load your content...
Please wait while we load your content...