A non-enzymatic electrochemical biosensor based on Au@PBA(Ni–Fe):MoS2 nanocubes for stable and sensitive detection of hydrogen peroxide released from living cells†
Abstract
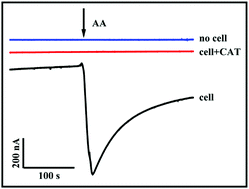
Hydrogen peroxide (H2O2) is the main product of enzymatic reactions and plays an important role in biological processes. The detection of H2O2 inside organisms or cells is critical. Here, we report a nickel–iron Prussian blue analogue nanocube doped with molybdenum disulfide and Au nanoparticles (Au@PBA(Ni–Fe):MoS2) as an electrochemical sensing material for the stable detection of H2O2 in neutral solutions for a long time. First, the Prussian blue analogue (PBA(Ni–Fe)) is synthesized by a simple charge-assembly technology, and then etched into PBA(Ni–Fe):MoS2 hollow nanocubes by a high-temperature hydrothermal reaction. Finally, Au nanoparticles are reduced inside the PBA(Ni–Fe):MoS2in situ to generate Au@PBA(Ni–Fe):MoS2 nanocubes. Ni-doping enhances the nanocube's stability in neutral solutions; as a result, the sensor can maintain a stable current response towards H2O2 reduction for more than 1 h. The sensing material can meet the needs of a long-time test. The introduction of Au enhances the electron transfer efficiency, which endows the sensor with good reduction ability for H2O2 at 0 V over a wide linear range (0.5–200 μM and 210–3000 μM) and with a low detection limit (0.23 μM (S/N = 3)), which fulfills the requirements for the detection of H2O2 in a biological system. The sensor can sense H2O2 released from cells stimulated by ascorbic acid. Au@PBA(Ni–Fe):MoS2 provides good guidance for the future development of efficient biosensors to be applied in cell biology.


 Please wait while we load your content...
Please wait while we load your content...