Spermine amides of selected triterpenoid acids: dynamic supramolecular system formation influences the cytotoxicity of the drugs†
Abstract
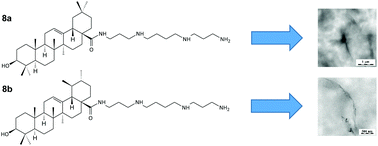
Cancer is a global disease of great importance, and the need for novel cytotoxic drugs is still eminent. A series of spermine amides of several selected triterpene acids (betulonic, heterobetulonic, oleanolic, ursolic and platanic acid) have been synthesized to search for new cytotoxic and antimicrobial agents. The compounds have also been subjected to the investigation of their physico-chemical characteristics (ability to self-assemble), and to an in silico comparative calculation of their physico-chemical and ADME parameters. In the in vitro screening tests with several target compounds (8a–8c and 11c), their cytotoxicity changed with prolonged time, which appeared to be a result of formation of dynamic supramolecular networks. This phenomenon is important in investigation of the effect of self-assembly on biological activity. The most important compounds in this series were spermine derivatives of heterobetulonic acid (3b) and ursolic acid (8b), showing cytotoxicity <5 μM and <10 μM, respectively, on all tested cancer cell lines. Comparable cytotoxicity was also displayed by 13b, formerly a model compound prepared for testing of the synthetic procedures, the 1,2-diaminoethane derivative. The target compounds 3b and 8b displayed antimicrobial activity on Staphylococcus aureus, Streptococcus mutans and Listeria monocytogenes at a concentration 6.25 μM. Supramolecular characteristics of several compounds were documented by the TEM and SEM micrographs showing fibrous, partially helical, networks, and UV measurements showing changes in the intensity of UV signals, also indicating formation of supramolecular systems.


 Please wait while we load your content...
Please wait while we load your content...