Fundamentals of cross-seeding of amyloid proteins: an introduction
Abstract
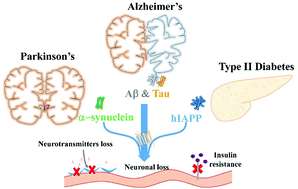
Misfolded protein aggregates formed by the same (homologous) or different (heterologous/cross) sequences are the pathological hallmarks of many protein misfolding diseases (PMDs) including Alzheimer's disease (AD) and type 2 diabetes (T2D). Different from homologous-amyloid aggregation that is solely associated with a specific PMD, cross-amyloid aggregation (i.e. cross-seeding) of different amyloid proteins is more fundamentally and biologically important for understanding and untangling not only the pathological process of each PMD, but also a potential molecular cross-talk between different PMDs. However, the cross-amyloid aggregation is still a subject poorly explored and little is known about its sequence/structure-dependent aggregation mechanisms, as compared to the widely studied homo-amyloid aggregation. Here, we review the most recent and important findings of amyloid cross-seeding behaviors from in vitro, in vivo, and in silico studies. Some typical cross-seeding phenomena between Aβ/hIAPP, Aβ/tau, Aβ/α-synuclein, and tau/α-synuclein are selected and presented, and the underlying specific or general cross-seeding mechanisms are also discussed to better reveal their sequence–structure–property relationships. The potential use of the cross-seeding concept to design amyloid inhibitors is also proposed. Finally, we offer some personal perspectives on current major challenges and future research directions in this less-studied yet important field, and hopefully this work will stimulate more research to explore all possible fundamental and practical aspects of amyloid cross-seeding.
- This article is part of the themed collection: Recent Review Articles


 Please wait while we load your content...
Please wait while we load your content...