Mitochondria-targeted cationic liposomes modified with alkyltriphenylphosphonium bromides loaded with hydrophilic drugs: preparation, cytotoxicity and colocalization assay†
Abstract
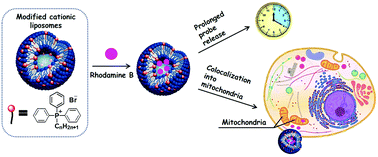
The purpose of this work was to obtain cationic liposomes based on 1,2-dipalmitoyl-sn-glycero-3-phosphocholine noncovalently modified using alkyltriphenylphosphonium bromides (TPPB-n) with different lengths of hydrocarbon tail for targeted delivery to mitochondria. The hydrodynamic diameter and electrokinetic potential of hybrid liposomes depending on the lipid/surfactant ratio were monitored in time with the aim to optimize the composition with sufficient stability and positive charge for mitochondria-targeted delivery. It was found that increasing the alkyl tail length of the surfactant (up to TPPB-14) leads to an increase in the positive charge of the liposomes. The most optimal results of stability were obtained for hybrid liposomes based on 1,2-dipalmitoyl-sn-glycero-3-phosphocholine and TPPB-12, TPPB-14. The obtained modified liposomes were loaded with hydrophilic substrates (a model probe Rhodamine B and medicines metronidazole and doxorubicin). This is one of the first examples of fabrication of liposomes noncovalently modified using an amphiphilic TPP cation, with the alkyl tail length of surfactant and TPP/lipid ratio optimized in terms of stability of the liposomes and the binding/release behavior of hydrophilic probes. Using the confocal microscopy method, it was shown that modification of liposomes with a triphenylphosphonium cation results in targeted delivery of encapsulated compounds to mitochondria.


 Please wait while we load your content...
Please wait while we load your content...