A self-assembled RNA-triple helix hydrogel drug delivery system targeting triple-negative breast cancer†
Abstract
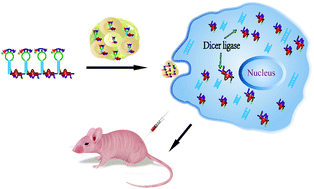
The major drawbacks of traditional RNA cancer therapies include low cellular uptake in vitro or in vivo, instability of in vivo circulation, nonspecific bio-distribution, and lack of targeting ability, which result in poor silencing efficiency. Herein, we developed a novel RNA-triple-helix hydrogel for the treatment of triple negative breast cancers (TNBCs) by incorporating RNA-triple-helix and siRNA duplexes of CXCR4 into the same RNA nanoparticles with no synthetic polycationic reagents added. The RNA-triple-helix consists of one tumour suppressor miRNA (miRNA-205) and one oncomiR inhibitor (miRNA-221), both of which showed an outstanding effect in synergistically abrogating tumours. The siRNA duplexes of CXCR4 were embedded into the RNA hydrogel to block breast cancer metastasis and conjugation of the LXL-DNA aptamer (apt-DNA-Chol) is an effective target DNA sequence for MDA-MB-231 cells. The self-assembly of the RNA-triple-helix hydrogel exhibited high selectivity of in vitro and in vivo absorption and controlling miRNA expression when compared to free miRNA and RNA transcripts. The well-developed gene delivery system provided a potential treatment with high specificity and selectivity toward TNBCs. This strategy can be implemented in triplex-helix hydrogel design to form novel miRNA combinations to treat various human cancers.
- This article is part of the themed collections: 2020 Journal of Materials Chemistry B most popular articles, Journal of Materials Chemistry B Lunar New Year collection 2021 and Biosensors


 Please wait while we load your content...
Please wait while we load your content...