Multifunctional fibrous scaffolds for bone regeneration with enhanced vascularization†
Abstract
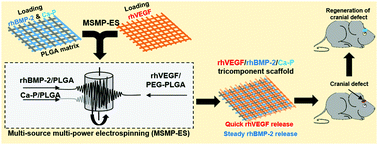
Due to the structural similarity to the extracellular matrix of human tissue and the ultra-high surface area-to-volume ratio, three dimensional electrospun fibrous structures have been increasingly used as tissue engineering scaffolds. Given that successful bone regeneration requires both good osteogenesis and vascularization, producing scaffolds that have both osteogenic and angiogenic potential is highly desirable. In this investigation, tricomponent fibrous scaffolds simultaneously incorporated with recombinant human vein endothelial growth factor (rhVEGF), recombinant human bone morphogenetic protein-2 (rhBMP-2) and bioactive calcium phosphate (Ca-P) nanoparticles are produced through a novel multi-source multi-power electrospinning method, and sequential growth factor release with a quick rhVEGF release and a steady rhBMP-2 release is achieved. The enhanced human umbilical vein endothelial cell (HUVEC) migration and tube formation, and up-regulated human bone marrow derived mesenchymal stem cell (hBMSC) osteogenic differentiation and mineralization demonstrate that tricomponent scaffolds have balanced angiogenic–osteogenic properties in vitro. 8 weeks after the scaffold implantation into the cranial defects of mice, obvious new bone regeneration and newly formed capillaries are observed in tricomponent scaffolds, suggesting that the tricomponent scaffolds enhance osteogenesis in vivo with required vascularization, which shows the great potential of the tricomponent scaffolds in bone tissue regeneration.


 Please wait while we load your content...
Please wait while we load your content...