Dually optimized polycaprolactone/collagen I microfiber scaffolds with stem cell capture and differentiation-inducing abilities promote bone regeneration
Abstract
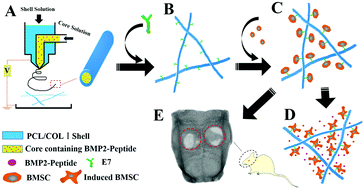
Micro–nano based fibrous scaffolds have been extensively studied in regenerative medicine. Bone marrow stem cells (BMSCs) and BMP2-derived peptides, two other important components for tissue engineering, have been successfully used for bone regeneration. However, a scaffold that specifically captures BMSCs and delivers BMP2-derived peptides to promote osteogenic differentiation of enriched BMSCs has not been reported. In this study, a microfiber scaffold was constructed by coaxial electrospinning technology using a polyvinylpyrrolidone/bovine serum albumin/BMP2-derived peptide compound as the core solution and a polycaprolactone/collagen I compound as the shell solution. The scaffolds were further functionalized by covalent grafting of a BMSC affinity peptide (E7) to develop a dual drug release system for the delivery of the BMP2-derived peptide and E7. Structural analysis indicated that the microfibers had a uniform diameter and homogeneous core–shell structure. Fourier transform infrared spectroscopy (FTIR) revealed that E7 was covalently bonded onto the surface of the fibers. In vitro, the E7-modified scaffolds promoted the initial adhesion of BMSCs and were more favorable for BMSC survival. Furthermore, the BMP2-derived peptide loaded in the E7-modified scaffolds was released in a sustained manner and retained bioactivity, significantly improving the osteogenic differentiation of BMSCs. In vivo, scaffolds loaded with the BMP2-derived peptide and E7 (PCME scaffolds) led to enhanced new bone formation and defect closure in a rat calvarial defect model. Overall, the PCME scaffold simultaneously facilitated all three of the essential elements needed for bone tissue engineering, providing a promising method for bone regeneration.


 Please wait while we load your content...
Please wait while we load your content...