Structural transitions and encapsulation selectivity of thermoresponsive polyelectrolyte complex micelles†
Abstract
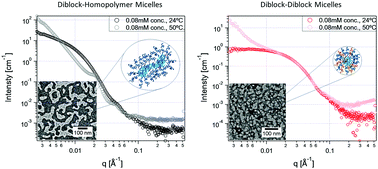
Polyelectrolyte complex (PEC) micelles are formed by mixing a block copolymer composed of a neutral block and a charged block, with an oppositely charged polymer. The micelles formed have a PEC core, capable of encapsulating charged molecules like nucleic acids or proteins, while the neutral block(s) forms the corona that offers protection that can prevent cargo from being degraded under physiological conditions. This work explores using a thermosensitive polymer, poly(N-isopropyl acrylamide) (pNIPAM), as the primary corona-forming block and how that can be leveraged in the context of drug delivery. pNIPAM has a lower critical solubility temperature (LCST), above which a hydrophilic to hydrophobic transition occurs. We are characterizing micelles formed using a diblock copolymer of pNIPAM–b-poly(acrylic acid) with (1) poly(lysine) and (2) poly(ethylene glycol)-b-poly(lysine) using dynamic light scattering, small angle X-ray scattering, absorbance and fluorescence spectroscopy, and transmission electron microscopy. Our results indicate that mixing pNIPAM–b-poly(acrylic acid) and poly(lysine) creates worm-like micelles, while mixing the same diblock with poly(ethylene glycol)-b-poly(lysine) forms spherical micelles. At temperatures above 35 °C, the transition temperature of pNIPAM–b-poly(acrylic acid), the worm-like micelles lose their structure, while the spherical micelles retain their structure, but both aggregate into larger assemblies. Lastly, we evaluate the ability of these micelles to encapsulate and release model charged therapeutics, using a cationic monoion, methylene blue and a cationic polyion, polylysine conjugated rhodamine. We find that methylene blue is not encapsulated by the micelle, and instead exhibits counterion-like behaviour upon polyelectrolyte complex formation. Conversely, fluorescence measurements of rhodamine-labelled polylysine show fluorescence quenching indicating that the polyion is encapsulated. By mixing quenched and unlabelled micelles, we show that both systems are immune to molecular exchange with their environment and instead the cargo remains trapped in the quenched micellar cores. Measurement of the micelle fluorescence above the LCST, decreases indicating no substantial release for either system. However, the increase in micelle quenching above the LCST and its persistence after cooling may offer an additional protective environment for cargo that can be triggered by temperature.
- This article is part of the themed collection: Journal of Materials Chemistry B Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...