Well-defined biological sample-compatible molecularly imprinted polymer microspheres by combining RAFT polymerization and thiol–epoxy coupling chemistry†
Abstract
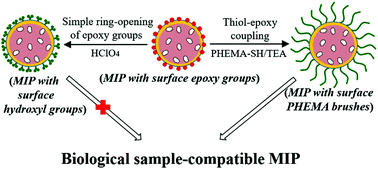
Molecularly imprinted polymers (MIPs) capable of selectively recognizing small organic analytes in complex biological samples hold great promise in many real-world bioanalytical and biomedical applications, but development of such advanced synthetic receptors remains a challenging task. Herein, a facile and highly efficient new approach to obtaining well-defined complex biological sample-compatible MIP microspheres is developed by combining RAFT polymerization and thiol–epoxy coupling chemistry. Its proof-of-principle has been demonstrated by the first synthesis of propranolol-imprinted polymer microspheres with surface epoxy groups (briefly MIP-EP) via the combined use of reversible addition–fragmentation chain transfer (RAFT) precipitation polymerization and surface-initiated RAFT polymerization and their subsequent coupling reaction with a hydrophilic macromolecular thiol (i.e., thiol-terminated poly(2-hydroxyethyl methacrylate) (PHEMA-SH)). MIP-EP proved to show good propranolol recognition ability in an organic solvent but not in aqueous solution. The coupling reaction between PHEMA-SH and MIP-EP readily led to MIP microspheres with densely grafted PHEMA brushes, which greatly enhanced the surface hydrophilicity of MIP particles and transformed water-incompatible MIP-EP particles into biological sample-compatible ones (i.e., the resulting hydrophilic MIP microspheres exhibited specific template binding almost as good as they showed in the organic solvent and high template selectivity in biological samples including undiluted pure milk and pure bovine serum). In sharp contrast, the simple ring-opening of the epoxy groups on MIP-EP particles by using perchloric acid (following a previously reported method for obtaining water-compatible MIPs) only provided MIPs with propranolol recognition ability in pure water instead of in the complex biological samples.


 Please wait while we load your content...
Please wait while we load your content...