Room-temperature synthesized porous Cu(OH)2/Cu7S4 hybrid nanowires as a high-performance electrode material for asymmetric supercapacitors†
Abstract
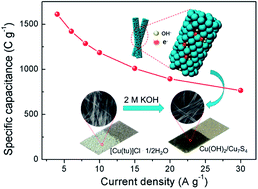
Copper-based nanomaterials have attracted significant interest as electrode materials for supercapacitors due to their low cost and environmental friendliness, however, their low capacitances inhibit their commercial applications. Design and application of porous and hybrid 1-D nanostructures as electrode materials is an effective approach to improve their capacitance. In this work, porous Cu(OH)2/Cu7S4 hybrid nanowires were synthesized at room temperature using a self-sacrificial template method. They had large surface areas and pore volumes, which provided numerous active sites for faradaic reactions and facilitated fast ion diffusion and electron transport during the charge–discharge process. Cu(OH)2/Cu7S4 nanowires coated on nickel foam were used as electrodes without using any conductive agent or binder. Results showed that a high specific capacity of 1610.8 C g−1 was achieved at a current density of 4 A g−1, and a large value of 765.0 C g−1 was still retained at a higher current density of 30 A g−1. An asymmetric supercapacitor assembled using this Cu(OH)2/Cu7S4 positive electrode and an active carbon negative electrode demonstrated a high energy density of 48.1 W h kg−1 at a power density of 429.3 W kg−1 and a good cycling stability, suggesting that these porous Cu(OH)2/Cu7S4 hybrid nanowires are promising electrode materials for supercapacitors.


 Please wait while we load your content...
Please wait while we load your content...