In situ engineered ZnS–FeS heterostructures in N-doped carbon nanocages accelerating polysulfide redox kinetics for lithium sulfur batteries†
Abstract
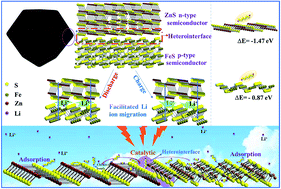
Electrode materials with efficient catalyzing capability for polysulfides in lithium sulfur batteries are currently receiving intensive research interest for next-generation portable electronic equipment. Herein, a novel hollow architecture composed of ZnS–FeS heterostructures encapsulated in N-doped carbon is designed for the first time as a high-efficiency catalyst to propel polysulfide redox kinetics, in which the ZnS–FeS heterostructures are mosaiced in the carbon framework through a simple in situ sulfuration process. Kinetic analyses and theoretical calculation verify that the abundant heterojunctions could facilitate electron and ion transfer, strengthen the combination with polysulfides and boost the polysulfide redox reaction kinetics. Ex situ electrochemical impedance spectroscopy (EIS) revealed the excellent interface solid–liquid–solid conversion reaction. Benefiting from the state-of-the-art design, the S@ZnS–FeS@NC electrode shows an outstanding rate capacity (718 mA h g−1 at 4.0C) and favorable cycling stability (822 mA h g−1 at 0.2C after 200 cycles). Our approach would be a proof-of-concept design of metal sulfide heterojunctions as an effective sulfur host in improving the polysulfide redox kinetics for lithium sulfur batteries.


 Please wait while we load your content...
Please wait while we load your content...