Unveiling the role of hydrothermal carbon dots as anodes in sodium-ion batteries with ultrahigh initial coulombic efficiency†
Abstract
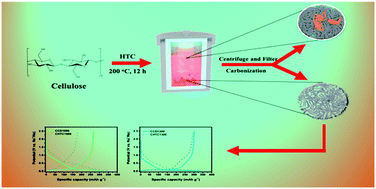
Hard carbon materials are regarded as the most promising anode materials for sodium-ion batteries (SIBs) due to their best cost-effectiveness. However, their relatively low specific capacity and initial Coulombic efficiency (ICE) compared with the graphite anode in lithium-ion batteries still limit the energy density for further development. Thus, it is necessary to produce high-performance hard carbon anode materials with high ICE to improve the SIB technology. Here we show the use of usually ignored carbon dots from the supernatant of hydrothermal carbonization (HTC) as anodes for SIBs after directly drying and carbonization. Compared to traditional HTC carbon spheres from the solid phase, the further carbonized carbon dots exhibit an excellent specific capacity of over 300 mA h g−1 with a significantly enhanced ICE of up to 91% at 30 mA g−1 which is among the highest values reported for carbonaceous anodes in SIBs. The superior ICE could benefit a high energy density of 248 W h kg−1 in full cells with the NaNi1/3Fe1/3Mn1/3O2 cathode. This new discovery from the simple traditional method provides new aspects of designing high-performance SIBs in future commercialization.


 Please wait while we load your content...
Please wait while we load your content...