High-performance alcohol electrooxidation on Pt3Sn–SnO2 nanocatalysts synthesized through the transformation of Pt–Sn nanoparticles†
Abstract
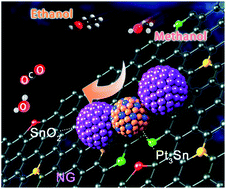
In this work, Pt3Sn–SnO2 supported on N-doped graphene (Pt3Sn–SnO2/NG) was synthesized through the in situ transformation of Pt–Sn/NG by annealing in an air atmosphere. The Pt3Sn–SnO2/NG catalyst, characterized by XRD, TEM and HRTEM, forms an ordered Pt3Sn intermetallic in which every Pt3Sn nanoparticle (NP) is in close contact with one or more SnO2 NPs. The Pt3Sn–SnO2/NG catalyst shows outstanding performance towards the ethanol oxidation reaction (EOR) and methanol oxidation reaction (MOR). The mass activity for EOR on the Pt3Sn–SnO2/NG catalyst is 469 mA mgPt−1 at 0.7 V, which is more than 10 times that of the commercial Pt/C catalyst (44 mA mgPt−1). The Pt3Sn–SnO2/NG catalyst is found to have more than three times the stability of the commercial Pt/C. Our Pt3Sn–SnO2/NG catalyst also delivers increased activity and higher stability for MOR compared with the Pt/C catalyst. In addition, the Pt3Sn–SnO2/NG catalyst is observed to have a higher CO tolerance compared with Pt/C. The high activity and stability of the Pt3Sn–SnO2/NG catalyst can be attributed to the unique structure of Pt3Sn–SnO2, formed by the in situ transformation of Pt–Sn/NG, which induces the formation of a small-sized ordered Pt3Sn intermetallic that strongly interacts with SnO2. The synergetic effect between the ordered Pt3Sn and SnO2 enhances the activity towards EOR and MOR through the Sn in both Pt3Sn and SnO2, which facilitates the removal of COads on the adjacent Pt active sites at low potentials by providing OHads species; it also improves stability because of the strong Pt3Sn–SnO2 interactions.


 Please wait while we load your content...
Please wait while we load your content...