Unveiling the role of tetragonal BiVO4 as a mediator for dual phase BiVO4/g-C3N4 composite photocatalysts enabling highly efficient water oxidation via Z-scheme charge transfer†
Abstract
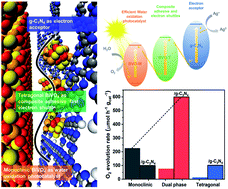
Monoclinic scheelite BiVO4 (BVO-M) based materials are of great interest as photocatalysts for water oxidation essential to produce value-added chemical fuels, but their slow charge transfer and low activity are yet to be resolved. As a solution to overcome this challenge, we report Z-scheme photocatalyst fabricated by integrating the dual phase structure of monoclinic scheelite and tetragonal zircon-type BiVO4 (BVO-T) moieties with graphitic carbon nitride (g-C3N4) having π-conjugated two dimensional layers suited to visible light-induced charge transfer. Moreover, V K-edge and Bi L3-edge X-ray absorption near-edge structure (XANES) and Fourier transformed extended X-ray absorption fine structure (EXAFS) spectra demonstrate that the tetragonal zircon-type BiVO4 structures in a Z-scheme catalyst form a bridge between BVO-M and g-C3N4 structures so that it acts as an electron mediator to accelerate charge transfer, in agreement with the zeta-potential analysis and the band structure revealed by UV-vis spectroscopy and ultraviolet photoelectron spectroscopy analyses. Furthermore, a Z-scheme photocatalyst is exhibited to show a 3-fold longer charge carrier lifetime than BVO-M, thereby enabling the greater than three-fold enhancement in photocatalytic water oxidation activity. Additionally, isotope 18O-labelling experiments reveal that evolved oxygen molecules result from water through photocatalytic water oxidation.


 Please wait while we load your content...
Please wait while we load your content...