Electrochemical reduction of N2 to ammonia on Co single atom embedded N-doped porous carbon under ambient conditions†
Abstract
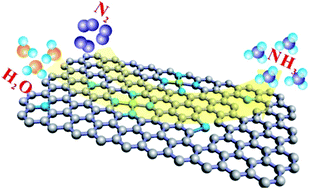
Energy-efficient ammonia synthesis from facile electrochemical N2 fixation is desirable in many fields such as fertilizer production and carbon-free energy carriers. However, it remains challenging due to the difficulty of N2 adsorption and dissociation. Here, Co single atom embedded N-doped porous carbon (CSA/NPC) was reported for electrochemical reduction of N2 to NH3, which exhibited high activity and efficiency at room temperature and atmospheric pressure. It can rapidly convert N2 to NH3 with an NH3 production rate of 0.86 μmol cm−2 h−1 and a faradaic efficiency of 10.5% at −0.2 V vs. (RHE). The Co single atoms, N-doping and porous structure were found to be responsible for its superior performance for ammonia synthesis, where Co single atoms and N-doping can provide active sites for N2 adsorption and dissociation and porous carbon can expose more active sites and enhance mass transfer.


 Please wait while we load your content...
Please wait while we load your content...