Ruthenium(iii) polyethyleneimine complexes for bifunctional ammonia production and biomass upgrading†
Abstract
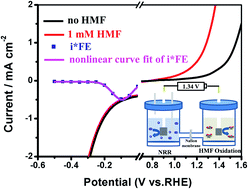
As an effective strategic approach to produce ammonia (NH3), electrocatalytic nitrogen reduction reactions (NRRs) under ambient conditions using renewable energy sources (e.g. solar) have attracted significant attention; however, the design of an efficient electrocatalyst for the NRR is a challenging task and has been of central research interest. Herein, we report the synthesis of ruthenium(III) polyethyleneimine (Ru(III)-PEI) catalysts supported on carboxyl-modified carbon nanotubes (Ru(III)-PEI@MWCNTs) by a self-assembly process driven by electrostatic forces at room temperature. Our newly designed Ru(III)-PEI@MWCNTs were employed as bifunctional catalysts for the NRR and 5-hydroxymethylfurfural (HMF) oxidation. At −0.10 V vs. the reversible hydrogen electrode (RHE), our Ru(III)-PEI@MWCNTs exhibited the high NH3 yield rate of 188.90 μgNH3 mgcat.−1 h−1 and the faradaic efficiency (FE) of 30.93% at room temperature. Furthermore, owing to its favorable thermodynamics for HMF oxidation, the Ru(III)-PEI@MWCNT electrode demonstrated an impressive electrocatalytic HMF oxidation at 1.24 V, 220 mV lower than that for oxygen evolution. The two-electrode electrolyzer employing Ru(III)-PEI@MWCNTs as a bifunctional catalyst for both the cathode and the anode showed the current density of 0.50 mA cm−2 with the cell voltage of only 1.34 V over 27 hours of stable electrolysis with a 94% FE for 2,5-furandicarboxylic acid (FDCA) production; this suggested an outstanding performance of this electrolyzer for the coupling of NRR with HMF oxidation. This study represents the first attempt at the ground demonstration of combining NH3 production with biomass upgrading.


 Please wait while we load your content...
Please wait while we load your content...