Pillar iodination in functional boron cage hybrid supramolecular frameworks for high performance separation of light hydrocarbons†
Abstract
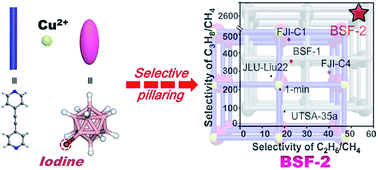
A novel ultramicroporous boron cage pillared supramolecular metal–organic framework BSF-2 comprising functional cluster undecahydro-iodo-closo-dodecaborate [B12H11I]2− pillars is designed and prepared by supramolecular assembly of CuB12H11I and 1,2-bis(4-pyridyl)acetylene (bpa). The structure and properties of BSF-2 are fully characterized by single crystal X-ray analysis, powder X-ray analysis, IR spectroscopy, EPR spectroscopy and TGA. The gas adsorption study of BSF-2 indicates substantially enhanced isosteric heats of adsorption (Qst) for C3H8, C2H6, C2H2 and CO2 but an almost identical Qst for CH4 as a result of adding an iodo group to the dodecaborate pillars. Moreover, the C3H8 capacity on BSF-2 is increased while the CH4 uptake capacity is significantly reduced compared to parent BSF-1, leading to excellent separation selectivities for C3H8/CH4, C2H6/CH4, C2H2/CH4 and CO2/CH4. The gas adsorption behavior of C3H8, C2H2 and CH4 in BSF-2 is further revealed by modeling studies, indicating an enhanced affinity of BSF-2 towards C3H8 and C2H2 but a weak affinity towards CH4, consistent with the experimental results. In addition, the practical separation of a ternary C3H8/C2H6/CH4 gas mixture is investigated by recyclable dynamic breakthrough experiments. This work sheds light on designing high performance microporous materials for gas separation and provides new insights into understanding hydrocarbon adsorption/separation in supramolecular metal–organic frameworks.


 Please wait while we load your content...
Please wait while we load your content...