Mesoporous palladium–boron alloy nanospheres†
Abstract
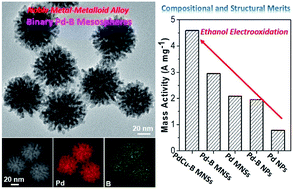
Noble-metal–metalloid binary palladium–boron (Pd–B) nanoalloys are interesting because the smaller boron atoms enlarge the Pd–Pd interlattice spacings and modify the binding energy barriers of catalytic intermediates. Binary Pd–B nanoalloys with nanostructured morphologies are an emerging class of (electro)catalysts that leverage these properties to enhance their performance in various chemical reactions. We describe here the first synthesis of Pd–B alloy mesoporous nanospheres (MNSs) and evaluate their electrocatalytic performance in the ethanol oxidation reaction (EOR). This method uses dimethylamine borane and boric acid as both the reducing agents and boron sources and amphiphilic dioctadecyldimethylammonium chloride (DODAC) as the surfactant template. The cylindrical mesophase of DODAC inhibits the mobility of the Pd metal precursor and confines the crystalline growth to form binary Pd–B MNSs with three-dimensional dendritic center-radial mesochannels. We demonstrate that the synthetic protocol can be adopted to rationally tune the diameters of the Pd–B MNSs from 30 nm to 120 nm without destroying the mesoporous structure and elemental composition. The Pd–B MNSs combine high surface area with favorable electrocatalytic surface properties to generate exceptional electrocatalytic performance for the EOR under alkaline conditions, illustrating the potential of this method as a platform to yield a new type of highly efficient electrocatalyst.


 Please wait while we load your content...
Please wait while we load your content...