Single atom-supported MXene: how single-atomic-site catalysts tune the high activity and selectivity of electrochemical nitrogen fixation†
Abstract
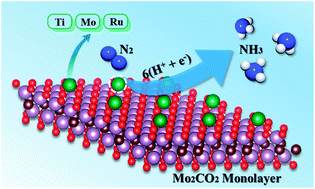
A central topic for the electrocatalytic N2 reduction reaction (ENRR) lies in the search for facile electrocatalysts under ambient conditions. Inspired by recent works on stabilizing single atoms with Ti3C2Tx MXene nanosheets, herein, we designed a series of single atoms supported on Ti2CO2 and Mo2CO2 MXene monolayers as efficient electrocatalysts for the ENRR by a well-defined first-principles calculation. Our calculation results revealed that single Ru or Mo atom anchored Mo2CO2 or Ti2CO2 possessed high ENRR activity, and the calculated limiting potentials of Ti2CO2 were more negative than those of Mo2CO2 due to the high conductivity of Mo2CO2. Moreover, N2 can be efficiently reduced to NH3 on Ti@Mo2CO2via different reaction mechanisms with a record limiting potential (−0.64 V). Furthermore, the activity and selectivity of the ENRR on Mo@Mo2CO2 were evaluated by comparing the Gibbs free energy of each dinitrogen and hydrogen as well as the first dinitrogen protonation and hydrogen adsorption. We found that N2 reduction proceeds via a distal or hybrid mechanism with an overpotential down to 0.16 or 0.19 V, indicating Mo@Mo2CO2 as a robust electrocatalyst for the ENRR. This work opens up an exciting new avenue for the rational design of high-efficiency MXene-based nanocomposites for artificial N2 fixation.


 Please wait while we load your content...
Please wait while we load your content...