Interfacial synergy of ultralong jagged Pt85Mo15–S nanowires with abundant active sites on enhanced hydrogen evolution in an alkaline solution†
Abstract
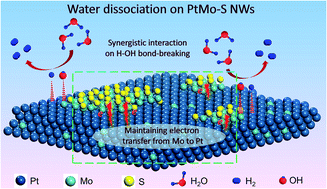
The higher electrocatalytic activity of hydrogen evolution in basic media for Pt-based catalysts is largely dependent on the intrinsic microscopic surface chemical environment. Given the fundamental mechanism of the interfacial interaction between Pt and oxophilic components, engineering a moderate oxygen-friendly surface environment of Pt-based catalysts is beneficial for endowing Pt with high catalytic activity for the hydrogen evolution reaction (HER). Herein, we fabricated ultralong jagged Pt85Mo15–S nanowires with rich interfacial active sites by using S as the “active auxiliary” to demonstrate an enhanced catalytic HER performance. Strikingly, the Pt85Mo15–S nanowires exhibited exceptional activity with 3.62 times the specific current density and 4.03 times the mass current density of commercial Pt/C as well as excellent stability towards alkaline HER. X-ray photoelectron spectroscopy revealed that the S element could prevent the further oxidization of Mo and then lead to enough electron transfer from Mo to Pt. In addition, the theoretical calculations demonstrated that the water dissociation energy barrier could be significantly reduced under the interfacial synergy of the intimate contact between Pt and MoSx. This work illustrates a new strategy to advance the fundamental exploration on electrocatalytic materials, which makes a systematic approach to build an interfacial model catalyst for fundamental electrocatalytic studies of alkaline HER.
- This article is part of the themed collection: International Year of the Periodic Table : Single Atoms as Active Catalysts


 Please wait while we load your content...
Please wait while we load your content...