Effective oxygen reduction on A-site substituted LaCuO3−δ: toward air electrodes for SOFCs based on perovskite-type copper oxides†
Abstract
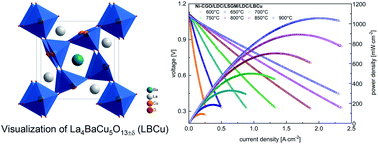
It is shown that appropriately doped Cu-based perovskite-type oxides exhibit high catalytic activity toward the oxygen reduction reaction at high temperatures, surprisingly making them suitable candidates for the preparation of air electrodes for solid oxide fuel cells. The proposed substitution in the La-sublattice of LaCuO3−δ by Ba and/or Sr (i.e. La4BaCu5O13±δ, La3SrCu4O10±δ and La3Sr0.6Ba0.4Cu4O10±δ compositions) allows stabilization of the perovskite-type structure at ambient pressures in a wide range of temperatures. The materials exhibit extraordinarily high total electrical conductivity associated with the presence of mixed Cu3+/Cu2+ states, limited thermal expansion due to suppressed oxygen release at high temperatures, as well as suitable thermal and chemical stability. Moreover, as derived from first principles calculations for the La4BaCu5O13±δ-based supercell, the activation energy of migration of the oxygen vacancies for La4BaCu5O13−δ is low, ca. 0.4 eV. In contrast to Co, copper is cheap, environmentally benign and thermal treatment of its oxides does not require high temperatures, and therefore, the proposed Cu-based compounds are attractive for preparation of the electrode layers. For the selected La4BaCu5O13±δ oxide the influence of sintering temperature on the cathodic polarization resistance is presented, with the lowest recorded value of 0.03 Ω cm2 at 900 °C. The performance of a button-type solid oxide fuel cell with the optimized La4BaCu5O13±δ-based air electrode screen-printed on the La0.2Ce0.8O2−δ buffer layer and La0.8Sr0.2Ga0.8Mg0.2O3−δ solid electrolyte is good, with the highest power output exceeding 1 W cm−2 at 900 °C for the cell fuelled with wet H2.


 Please wait while we load your content...
Please wait while we load your content...