Highly dispersive and stable Fe3+ active sites on 2D graphitic carbon nitride nanosheets for efficient visible-light photocatalytic nitrogen fixation†
Abstract
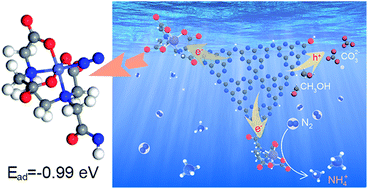
As an indispensable chemical, ammonia is produced by the Haber–Bosch process using H2 and N2 under severe reaction conditions. Recently, photocatalysis using solar energy has been regarded as a promising technology to fix N2 under mild conditions. However, compared to other photocatalytic applications, photocatalytic N2 fixation suffers from low efficiency because of the difficulties associated with N2 adsorption and activation on the photocatalyst surface, as well as the instability of active sites on the photocatalyst. Herein, we covalently grafted an ethylenediaminetetraacetic acid (EDTA) motif onto graphitic carbon nitride nanosheets (CNNS) by a high-temperature dehydration reaction, and subsequently chelated Fe3+ ions to obtain highly dispersive and stable Fe3+ sites on CNNS. Compared with the un-modified sample, the obtained Fe-EDTA–CNNS has improved visible light absorption and high charge separation and transfer ability. Impressively, a much higher activity for N2 fixation (50 μmol L−1 h−1) than those of the comparative materials and photocatalysts in the literature as well was achieved by the Fe-EDTA–CNNS. Experimental and calculation results demonstrate that the modification of CNNS with an Fe-EDTA unit enhances the reduction ability of photo-generated electrons and decreases the N2 adsorption energy. More importantly, the Fe-EDTA unit with an electron-withdrawing effect can accumulate photo-excited electrons and then activate the adsorbed N2 molecules. Additionally, the strong chelating action results in a relatively high stability for the Fe-EDTA–CNNS photocatalyst. This work not only develops a potential photocatalyst for efficient ammonia production, but also provides a facile surface modification strategy to construct active sites and regulate the charge transfer route on photocatalysts.


 Please wait while we load your content...
Please wait while we load your content...