Sandwich structured WO3 nanoplatelets for highly efficient photoelectrochemical water splitting†
Abstract
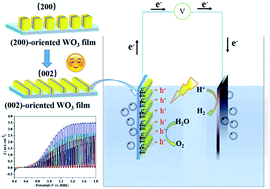
The most critical aspect for photoelectrochemical (PEC) water splitting is the choice of suitable photoelectrode materials. The low efficiency of PEC water splitting photoanodes restricts their widespread deployment for large scale H2 production. Herein, WO3 with controlled structures was in situ hydrothermally grown on fluorine-doped tin oxide (FTO) glass substrates via simply using different orientation agents, i.e., citric acid for the sandwich structure and tartaric acid for nanoplates. By determining the electrochemically active surface area (ECSA), the sandwich structure is proven to be able to provide more active sites and a larger specific reaction surface area for PEC water splitting. In the citric acid derived WO3 sandwich structure, upon increasing the hydrothermal reaction time from 4 to 16 h, the dominating exposed facets were converted from the (200) crystal plane to the (002) crystal plane, while WO3 prepared from tartaric acid consistently yielded the (200) crystal plane. Moreover, the sandwich structured WO3 photoanode with intensively exposed (002) facets obtained hydrothermally in 12 h exhibited a photocurrent density of 3.16 mA cm−2 in aqueous Na2SO4 solution under AM 1.5 G illumination and an incident photon-to-current efficiency (IPCE) value of 79% at 400 nm at 1.23 V vs. RHE. To the best of our knowledge, this value is one of the most remarkable for pristine WO3 photoanodes. The excellent PEC performances of the resulting sample could be attributed to the 2-D nanoplates with superior crystallinity and exposed highly reactive (002) facets which facilitated the separation of photogenerated electron–hole pairs.


 Please wait while we load your content...
Please wait while we load your content...