Spatially controlled synthesis of superlattice-like SnS/nitrogen-doped graphene hybrid nanobelts as high-rate and durable anode materials for sodium-ion batteries†
Abstract
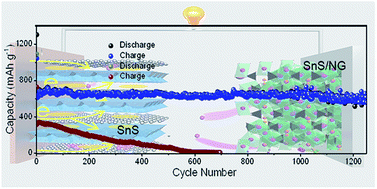
Sodium-ion batteries (SIBs) are promising for large-scale energy storage because of the low cost and earth abundance of sodium. Although tin monosulfide (SnS) has attracted broad interest in SIBs due to its layered structure and large capacity, the sluggish sodiation kinetics, substantial volumetric variation, and electrochemical dissolution of intermediate polysulfides during cycling hinder its practical implementation. Herein, we report a spatially confined synthesis of superlattice-like SnS/N-doped graphene (NG) hybrids composed of alternately stacked SnS and NG nanosheets as high-rate and long-life anode materials for SIBs. The density functional theory calculations reveal the enhanced densities of states (DOSs) near the Fermi level and increased interfacial conductivity for the SnS/NG heterostructured nanobelts. In SnS/NG hybrids, the SnS is sandwiched between the NG layers enabling enlarged SnS interlayer spacing and a strong built-in field between adjacent SnS and NG, which facilitates the Na-ion diffusion and sodiation/desodiation reactions with rapid kinetics. Moreover, the conductive NG layers not only improve the conductivity in the composite but also provide the 2D confined interspace to accommodate the volume variation of SnS and mitigate polysulfide effusion and shuttling during cycling to yield excellent cycling stability. Consequently, the superlattice-like SnS/NG hybrid shows a large capacity of 600 mA h g−1 at 1600 mA g−1, a high rate capability of 550 mA h g−1 at 14 580 mA g−1 and an excellent cycling stability with 0.004% decay per cycle for over 1200 cycles, outperforming the reported sulfides so far. The full cell with the SnS/NG anode and Na3V2(PO4)3 cathode has a large energy density of 160 W h kg−1 and good cycling stability, demonstrating promising applications in electrochemical energy storage.


 Please wait while we load your content...
Please wait while we load your content...