Multi-layered photocathodes based on Cu2ZnSnSe4 absorber and MoS2 catalyst for the hydrogen evolution reaction†
Abstract
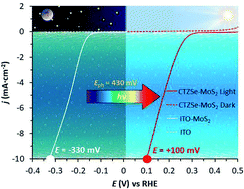
A multilayered p-type photoabsorber based on kesterite Cu2ZnSnSe4 has been functionalized with inexpensive and easily scalable amorphous MoS2 electrocatalyst by cathodic photoelectrodeposition. The resulting photocathodes with a final composition of Mo/MoSe2/Cu2ZnSnSe4/CdS/ZnO/ITO/MoS2 (where ITO = indium tin oxide) feature one of the highest hydrogen evolution catalytic current densities reported to date at a potential E = 0 V vs. RHE (−18 mA cm−2 in pH 2) under 1 sun illumination. A comparison of the performance of the photoelectrode with an analogous dark electrode with composition ITO/MoS2 allows us to estimate a photovoltage of 430 mV, which is very close to the working voltage of an equivalent photovoltaic cell with open circuit voltage VOC = 464 mV. The photoelectrodes show sustained activity at −10 mA cm−2 above the thermodynamic potential of the H+/H2 redox couple (E = +86 mV vs. RHE in pH 2) for 1 hour without any loss of activity. Prolonged reaction times up to 4 hours show no degradation of the photoabsorber multilayered architecture but a decrease in the thickness of the MoS2 electrocatalytic film due to the mechanical stress caused by the hydrogen bubbles forming on the surface of the electrode. The MoS2 layer is key not only to catalyze the catalytic reaction but also to protect the photoabsorber from the corrosive electrolyte solution. The adhesion of the MoS2 electrocatalytic film on the ITO top layer is critical for the stability during hydrogen evolution catalysis.


 Please wait while we load your content...
Please wait while we load your content...