Activating MoS2 basal planes for hydrogen evolution through direct CVD morphology control†
Abstract
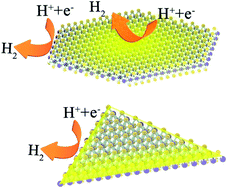
Monolayer MoS2 has emerged as an active and non-precious electrocatalyst for electrochemical hydrogen production. The atomic thinness and ultrahigh surface-to-volume ratio of the chemical vapor deposition (CVD)-grown monolayers result in an ideal material to facilitate efficient electrochemical hydrogen evolution and explore the mechanism at the atomic level. However, the active sites of pristine monolayer MoS2 are reported to locate at the edges, leaving the basal planes inert, which limits their hydrogen evolution reaction performance. Here, we synthesize monolayer MoS2 hexagonal flakes with high surface coverage and abundant highly distributed S vacancies as active reaction sites directly by CVD. The catalytic performance of the hexagonal MoS2 flakes presented here is superior to that of the existing as-grown MoS2, exhibiting a current density of 100 mA cm−2 at −353 mV versus the reversible hydrogen electrode (RHE) with an extraordinarily low onset potential of only 41 mV. And an outstanding exchange current density of 0.091 mA cm−2 stands as the highest ever reported for all kinds of non-precious-metal doped MoS2 catalysts. It is proved by a variety of tests that abundant S vacancies are distributed in the highly crystalline basal plane of hexagonal MoS2, which leads to a remarkably improved catalytic efficiency compared with that of the triangular one. The monolayer MoS2 is further evidenced to have excellent long-term stability and high durability, maintaining a stable performance for nine months in air and retaining the initial performance after 6000 cyclic voltammetry scans. On-site defect engineering of monolayer MoS2 for excellent hydrogen evolution reaction (HER) performance enriches insights into the structure–performance relationship in 2D materials and provides new opportunities for the design of highly active catalysts.


 Please wait while we load your content...
Please wait while we load your content...