A zinc(ii) complex of di(naphthylethynyl)azadipyrromethene with low synthetic complexity leads to OPV with high industrial accessibility†
Abstract
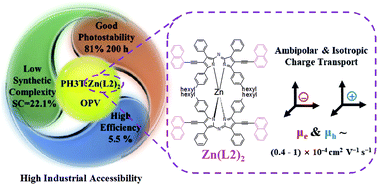
Organic photovoltaics have reached high power conversion efficiencies (PCE) using non-fullerene acceptors (NFAs). However, the best NFAs tend to have complex syntheses, limiting scalability. Among polymer donors, regioregular poly(3-hexylthiophene) (P3HT) has the greatest potential for commercialization due to its simple synthesis and good stability, but PCEs have been limited. It is thus imperative to find scalable NFAs that give high PCE with P3HT. We report a zinc(II) complex of di(naphthylethynyl)azadipyrromethene (Zn(L2)2) as a non-planar NFA that can be synthesized on the gram scale using inexpensive starting materials without chromatography column purification. The NFA has strong absorption in the 600–800 nm region. Time-dependent density-functional theory calculations suggest that the low-energy absorptions can be understood within a four-orbital model involving transitions between π-orbitals on the azadipyrromethene core. OPVs fabricated from P3HT:Zn(L2)2 blends reached a PCE of 5.5%, and the PCE was not very sensitive to the P3HT:Zn(L2)2 weight ratio. Due to its shape, Zn(L2)2 shows isotropic charge transport and its potential as an electron donor is also demonstrated. The combination of simple synthesis, good PCE and photostability, and tolerance to the active material weight ratio demonstrates the potential of Zn(L2)2 as an active layer material in OPVs.


 Please wait while we load your content...
Please wait while we load your content...