Ultra-small metal nanoparticles supported on carbon nanotubes through surface chelation and hydrogen plasma reduction for methanol electro-oxidation†
Abstract
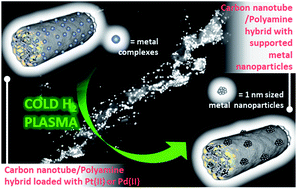
This paper reports a new method to obtain ultra-small Pd and Pt nanoparticles (0.5–1 nm) supported on multi-walled carbon nanotubes (MWCNTs). Even at high loadings of both metals (22.3 and 31.5% (wt/wt) of Pd2+ and Pt2+, respectively), very narrow and unimodal particle size distributions are achieved. The complexing capabilities of polyethyleneimine covalently attached to the surface of the tubes are optimal for the retention of Pd2+ and Pt2+ from solution. We have addressed the reduction of the retained ions by two approaches: a classical treatment with NaBH4 in aqueous solution, and a novel method using hydrogen cold plasma to preserve the structural features of the material. Cold plasma produced degrees of reduction similar or even larger than NaBH4, supporting the advantage of hydrogen cold plasma as reducing agent as it is a simple, clean and fast (15 minutes) procedure. XPS analysis of the reduced materials show an increase in the electron density near the Fermi level. Pt/MWCNT materials have been tested as anode for methanol electrooxidation, showing a catalytic profile typical of that observed for platinum nanoparticles. The stability after 1000 cycles of the plasma-reduced materials is much larger than these reported for Pt/carbon materials, indicating the stability of the ultra-small nanoparticles.


 Please wait while we load your content...
Please wait while we load your content...