Cu single atoms on Ti2CO2 as a highly efficient oxygen reduction catalyst in a proton exchange membrane fuel cell†
Abstract
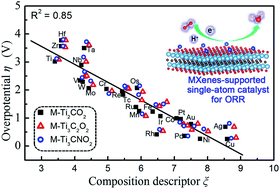
Although carbon-based single-atom catalysts (SACs), especially Fe–N–C, have been demonstrated as highly promising electrocatalysts for the oxygen reduction reaction (ORR) in alkaline media, their application remains a great challenge in acidic proton exchange membrane fuel cells (PEMFCs). Here, by performing high throughput first-principles calculations for 3d, 4d and 5d transition-metal single atoms immobilized on two-dimensional (2D) titanium carbide (Ti2CT and Ti3C2T) surfaces as active sites, we rationally design SACs towards a highly efficient ORR and further propose a composition descriptor to provide atomic-level insights into the structure–activity relationship. Significantly, the parameters involved in the present descriptor can be conveniently obtained from the periodic table of elements. More importantly, we found that the Ti2CO2-supported non-noble Cu SACs exhibit excellent ORR activity with much lower overpotential (0.25 V) than that of Pt/C (0.4 V), high selectivity for 4e oxygen reduction, excellent stability and acid-resistance and are quite promising ORR catalysts for PEMFCs. The present method can be extended to other 2D transition-metal carbides, i.e., MXenes.


 Please wait while we load your content...
Please wait while we load your content...