Multiple modulations of pyrite nickel sulfides via metal heteroatom doping engineering for boosting alkaline and neutral hydrogen evolution†
Abstract
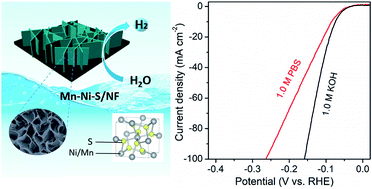
Rational design of alternative, cost-effective and highly active electrocatalysts for the hydrogen evolution reaction (HER) in alkaline and neutral media is greatly desirable and challenging. In this work, we developed a simple but effective manganese-metal-heteroatom doping strategy to realize the simultaneous modulations of the active site number, water dissociation, and hydrogen adsorption free energy in pyrite NiS2 hierarchical nanosheets to significantly boost alkaline and neutral HER catalysis. Specifically, the incorporation of Mn heteroatoms into the NiS2 system, as revealed by HRTEM, XPS, XANES spectra and theoretical studies, not only induce lattice distortions and defects for increasing the exposure of active sites, but also effectively optimize the electronic structure configuration of Ni sites, leading to optimal hydrogen adsorption free energy. In addition, the doped Mn heteroatom itself can act as a water-activated site to lower the energy barrier of water dissociation. As a result, the synergistic regulation of active sites and HER kinetics brings nearly 9-fold enhancement of alkaline HER activity for Mn-doped NiS2/Ni foam (NF) with a quite low overpotential of 71 mV to reach 10 mA cm−2 in 1 M KOH, which is among the most active HER electrocatalysts reported to date. Despite few reports about the effective neutral HER on transition-metal sulfides so far, a small overpotential of 84 mV at 10 mA cm−2 can be achieved in 1 M phosphate-buffered saline (PBS, pH 7). Furthermore, the Mn-doped NiS2/NF electrode also exhibits efficient and stable HER performances in near-neutral real seawater and has no obvious catalytic degradation after various extreme bending tests, verifying its high flexibility and robustness under severe conditions, which vastly broadens its application prospects.


 Please wait while we load your content...
Please wait while we load your content...