A self-reconstructed (oxy)hydroxide@nanoporous metal phosphide electrode for high-performance rechargeable zinc batteries†
Abstract
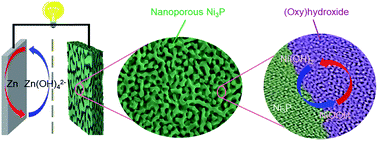
Rechargeable aqueous zinc batteries have attracted tremendous attention due to their remarkable high capacity, low cost, and good safety. However, their limited interfacial kinetics and poor rate performance have limited their application. Here, a new type of Ni//Zn rechargeable battery is fabricated with self-reconstructed (oxy)hydroxide@nanoporous Ni3P as a self-supported binder-free cathode. The (oxy)hydroxide@nanoporous Ni3P electrode exhibits an area specific capacity of 0.501 mA h cm−2 and a volume specific capacity of 167.0 mA h cm−3, and the alkali Zn battery achieves an energy density of 15.38 mW h cm−3 and a power density of 4.15 W cm−3, which are higher than most values reported in the literature. Benefiting from the good conductivity of the self-supported electrode, the efficient interfacial charge transfer of amorphous (oxy)hydroxide and fast ion diffusion via the pore channels in the binder-free electrode, the Ni//Zn battery exhibits excellent rate capability and cycling stability (94% retention after 6000 cycles). Combined with its good electrochemical stability and excellent rate performance, this novel electrode shows tremendous potential for application in high-capacity rechargeable Zn batteries.


 Please wait while we load your content...
Please wait while we load your content...