A two-dimensional semiconducting covalent organic framework with nickel(ii) coordination for high capacitive performance†
Abstract
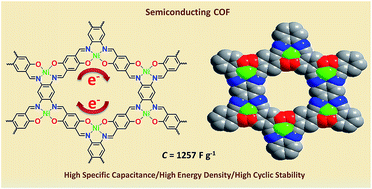
Covalent organic frameworks (COFs) have attracted enormous attention due to their unique structures, good crystallinity, modifiable functional groups, and excellent chemical stability. Nevertheless, their low electrical conductivity impeded their potential usage in supercapacitors. Herein, a two-dimensional conductive covalent organic framework (Ni-COF) with a square-planar Ni(II) coordination geometry is developed. The electrical conductivity of Ni-COF powder and thin film is 1.3 × 10−2 and 1.2 S cm−1, respectively. The highly conjugated skeleton, ordered porous structure and numerous redox centres contribute to a high specific capacitance of 1257 F g−1 at 1 A g−1 with 94% capacitance retention after 10 000 cycles. Meanwhile, the asymmetric supercapacitor (activated carbon//Ni-COF) shows a high capacitance of 417 F g−1 at 1 A g−1, and an outstanding energy density of 130 W h kg−1 at a power density of 839 W kg−1, outperforming previously reported COF electrode materials and even comparable to metal–organic framework systems. Thus, this study opens a new way to construct highly conductive structure-controllable COF electrode materials for supercapacitors.


 Please wait while we load your content...
Please wait while we load your content...