A solid-state dendrite-free lithium-metal battery with improved electrode interphase and ion conductivity enhanced by a bifunctional solid plasticizer†
Abstract
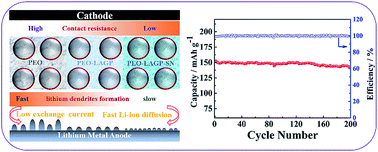
Solid-state electrolytes are promising candidates for applications in next-generation lithium-metal batteries (LMBs) because they eliminate the leakage hazards of liquid organic electrolytes and improve the reliability of LMBs in practical applications. However, the inherent defects such as low ionic conductivity and poor interfacial compatibility between the solid electrolyte and electrode hinder their extensive applications. At the same time, the rapid and uneven early nucleation of the lithium metal can lead to large amounts of dendrites in later stages, which gives rise to serious safety problems. Hence, a composite solid-state electrolyte, which consists of PEO, an inorganic ionic conductor, lithium aluminium germanium phosphate (LAGP), and a solid plasticizer, succinonitrile (SN), was explored to improve electrode interphase and ion conductivity. A Li-ion enrichment area could be formed by LAGP, indicating the formation of a fast Li-ion pathway. The crystallinity of PEO decreased effectively upon the introduction of LAGP and SN; thus, the optimized PEO18-LiTFSI-14 wt% LAGP-15 wt% SN electrolyte (SPE-14-15) exhibited excellent ionic conductivity of 1.26 × 10−4 S cm−1 at 30 °C. Meanwhile, SN could improve the interfacial compatibility between the electrode and SPE, therefore inhibiting the growth of lithium dendrites significantly. Li|Li symmetrical cells with the SPE-14-15 electrolyte yielded low hysteresis of 40 mV for 1000 h at 0.2 mA cm−2 and 40 °C. Notably, the Li|SPE|LFP all-solid-state LMB demonstrated outstanding specific capacity of 142.6 mA h g−1 after 200 cycles at 40 °C, and 91.2% of the initial capacity was retained. Moreover, for the first time, various electrochemical techniques combined with SEM analysis were performed to systematically investigate various chemical and kinetic properties of several composite SPEs on the deposition of lithium.


 Please wait while we load your content...
Please wait while we load your content...