Microparticles with hetero-nanointerfaces: controlled assembly of cobalt hydroxide and nickel hydroxide nanoclusters towards improved electrochemical functions†
Abstract
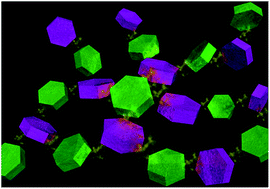
The ultimate control of the interfaces of nanocomposite materials is essential to tailor and improve their physical/chemical properties in applications such as catalysis and energy storage and production. Fabrication and co-assembly of a variety of nanostructured colloids are promising ways to design the interface of materials in the nano-scale toward high functionality. In this study, we demonstrate the synthesis of colloids of nanocluster-sized (∼2 nm) cobalt and nickel hydroxides and their assembly into microparticles that present hetero-nanointerfaces. The electrochemical properties were investigated to elucidate the effect of the hetero-nanointerface. Microparticles with hetero nanostructures composed of cobalt and nickel hydroxide nanoclusters revealed improved mass specific capacity (91.4 mA h g−1) compared with the respective microparticles with homo-nanointerfaces (cobalt hydroxide, 15.8 mA h g−1; nickel hydroxide, 64.4 mA h g−1). Further investigation suggests that the introduced hetero-nanointerface leads to lower charge transfer resistance and improved electrochemical properties. The synthetic concept demonstrated here is expected to create unique hetero-nanointerfaces for various materials with a wide-range of chemical compositions towards improved and novel functionalities.


 Please wait while we load your content...
Please wait while we load your content...