Stabilizing a sodium-metal battery with the synergy effects of a sodiophilic matrix and fluorine-rich interface†
Abstract
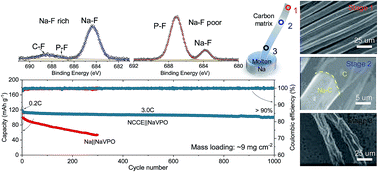
A sodium (Na) metal anode plays a pivotal role in realizing long-term cycling stability in promising Na-based batteries. However, the uncontrollable plating-stripping process and unstable solid electrolyte interphase (SEI) layer can induce electrode degradation, which hinders its potential applications. In this work, the synergy effects of a composite Na metal anode and fluorine-rich electrolyte were introduced to investigate the Na+ plating-stripping process and its potential use in Na-metal batteries (SMBs). The composite anode was fabricated via alloying and capillary action between the sodiophilic carbon matrix and molten Na metal, thus forming an integrated structure, which effectively improved the interconnection between Na and carbon scaffold to obtain uniform deposition. Combined with the fluorine-rich electrolyte, stable SEI layers consisting of ion-permeable NaF and mechanically durable organic components were obtained. When coupled with an Na3V2(PO4)3 cathode with a high mass loading of ∼9 mg cm−2, an ultra-stable cycling behaviour (capacity retention of >90% over 1000 cycles) and outstanding rate performance (capacity retention of >80% at 15C) were observed due to the improved Na deposition process and interface stability. This work demonstrates that the synergy effects of the composite metal anode and electrolyte modification can be an effective strategy to fabricate highly stable SMBs.


 Please wait while we load your content...
Please wait while we load your content...