Crystal size versus paddle wheel deformability: selective gated adsorption transitions of the switchable metal–organic frameworks DUT-8(Co) and DUT-8(Ni)†
Abstract
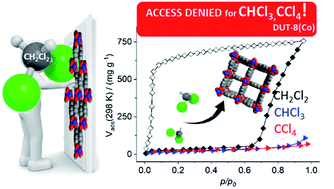
Switchable pillared layer metal–organic frameworks M2(2,6-ndc)2(dabco) (DUT-8(M), M = Ni, Co, 2,6-ndc = 2,6-naphthalenedicarboxylate, dabco = 1,4-diazabicyclo-[2.2.2]octane, DUT – Dresden University of Technology) were synthesised in two different crystallite size regimes to produce particles up to 300 μm and smaller particles around 0.1 μm, respectively. The textural properties and adsorption-induced switchability of the materials, obtained from both syntheses, were studied by physisorption of N2 at 77 K, CO2 at 195 K and n-butane at 273 K, revealing pronounced differences in adsorption behavior for Ni and Co analogues. While the smaller nano-sized particles (50–200 nm) are rigid and show no gating transitions confirming the importance of crystallite size, the large particles show pronounced switchability with characteristic differences for the two metals resulting in distinct recognition effects for various gases and vapours. Adsorption of various vapours demonstrates consistently a higher energetic barrier for the “gate opening” of DUT-8(Co) in contrast to DUT-8(Ni), as the “gate opening” pressure for Co based material is shifted to a higher value for adsorption of dichloromethane at 298 K. Evaluation of crystallographic data, obtained from single crystal and powder X-ray diffraction analysis, showed distinct geometric differences in the paddle wheel units of the respective MOFs. These differences are further disclosed by solid-state UV-vis, FT-IR and Raman spectroscopy. Magnetic properties of DUT-8(Co) and DUT-8(Ni) were investigated, indicating a high-spin state for both materials at room temperature. Density functional theory (DFT) simulations confirmed distinct energetic differences for Ni and Co analogues with a higher energetic penalty for the structural “gate opening” transformation for DUT-8(Co) compared to DUT-8(Ni) explaining the different flexibility behaviour of these isomorphous MOFs.


 Please wait while we load your content...
Please wait while we load your content...