Revealing structural evolution of PbS nanocrystal catalysts in electrochemical CO2 reduction using in situ synchrotron radiation X-ray diffraction†
Abstract
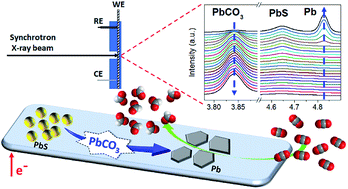
We investigated the structural evolution of PbS catalysts under electrochemical CO2 reduction reaction (eCO2RR) conditions using an in situ synchrotron radiation X-ray diffraction (SR-XRD) technique. The PbS catalyst with an initial form of size-tunable nanocrystals (NCs) performed efficiently in the electrochemical conversion of CO2 to formate with a record-high mass activity (MA) of 52.2 ± 5.2 mA mgPb−1 at −1.2 V and 74.9 ± 4.7 mA mgPb−1 at −1.4 V vs. the reversible hydrogen electrode (RHE). In situ SR-XRD analyses revealed that PbS NCs underwent rapid structural transformation to Pb thin films during the eCO2RR, providing the real catalytic surface for the eCO2RR. Such a structural change is complex, with PbS first being converted to PbCO3 and the latter species being further reduced to Pb. This work highlights the importance of understanding environmental impacts on the instability of nanocatalysts under eCO2RR conditions and the need for designing more robust electrocatalysts.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...