Self-supported copper-based gas diffusion electrodes for CO2 electrochemical reduction†
Abstract
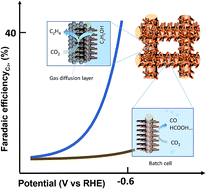
Nanostructured copper materials are catalytically active for the electrochemical reduction of CO2 (CO2RR) to produce hydrocarbons. However, most of these catalysts were investigated in conventional batch reactors at low current densities (<50 mA cm−2) due to the limitation of mass transport. Herein, a nondestructive strategy is proposed to transform nanostructured Cu catalysts into self-supported gas diffusion electrodes (GDEs), which enables the evaluation of the CO2 reduction performance in a flow cell at high current densities (up to 300 mA cm−2). Two typical self-supported GDEs were prepared through simply coating a hydrophobic microporous layer onto CuxO nanowires grown on Cu gauze. Both GDEs show high selectivity (>40%) for converting CO2 to multi-carbon products, e.g. C2H4 and ethanol, at commercially relevant current densities (>100 mA cm−2) and low overpotentials (η < 0.65 V). The GDEs are stable for more than 6 hours at current densities higher than 100 mA cm−2. Moreover, the nondestructive method allows us to directly compare the product distributions of the nanostructured Cu catalysts in a batch reactor and flow reactor and to demonstrate the influence of the reaction environment and mass transport on the CO2RR. While the CO2RR in the H-cell shows high selectivity towards CO and formate, the reaction in the flow cell produces a greater amount of multi-carbon products due to the fast CO2 diffusion and high pH. Combined, the electrode design strategies and the experimental findings presented in this work are valuable for the development of other self-supported electrodes for practical applications.


 Please wait while we load your content...
Please wait while we load your content...