Enhanced oxygen exchange capacity in nano-structured vanadia–ceria multi-phase oxygen carriers for solar thermal fuel production†
Abstract
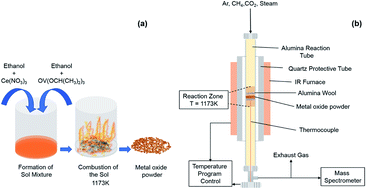
Developing an efficient redox material is a fundamental and crucial step in sustainable hydrocarbon fuel production via solar energy-driven thermochemical redox cycles. Vanadium being the 20th most abundant element in the earth's crust, with excellent catalytic properties combined with other oxide materials can potentially be a suitable candidate for efficient solar fuel production. Here, we report a first demonstration of a facile strategy to effectively utilize vanadium oxide with ceria for thermochemical H2O and CO2 splitting with superior oxygen exchange capacity and efficient H2 and CO yields of 68% more than the pure nano-structured ceria. We observe a synergic effect in oxygen exchange capacity and ion mobility in the vanadia–ceria binary phase system, where vanadia provides more reducing states for the hydrocarbon oxidation, while ceria acts as an oxygen buffer for the re-oxidation of reduced vanadia. We demonstrate that the addition of a carbon dioxide step between the methane partial oxidation and H2O splitting step activates the oxygen carrier by creating sufficient oxygen vacancies for the controlled oxidation of methane and avoiding methane cracking for over 100 cycles. The extraordinarily high oxygen exchange capacity observed in 25% V–CeO2 resulted in superior and pure H2 yields of 220 mL g−1 during the water splitting step of sequential water and carbon dioxide splitting, while maintaining a H2/CO ratio close to 2. Finally, these findings suggest that the facile combination of the extraordinary catalytic properties of vanadia and superior oxygen ion mobility of ceria can be a powerful approach for an efficient and effective solar thermochemical fuel production.


 Please wait while we load your content...
Please wait while we load your content...